 
The general formula for linear geometry is AX2, and thus CS2 shows linear geometry. Molecular geometryĪs the hybridization of CS2 is sp hybridization, the Carbon atom is in center bonding with two sulfur atoms forms the bond angle of 180 degrees, making the molecular geometry of CS2 molecule linear. Once you understand the hybridization of the molecule, it’s easy to determine the molecular geometry and the polarity of the molecule. Steric Number of CS2 is 2 thus its hybridization has two hybrid orbitals making it an sp hybridization. Here in the CS2 molecule, the number of sigma bonds on the central atom is two, and there are no lone pairs on the central atom as its octet is complete by sharing the valence electrons. = No of sigma bonds on the central atom +No of pi lone pairs on the central atom The formula to find the steric number for any molecule is: The steric number is the sum of the number of atoms bonded on the central atom and the number of lone pairs of electrons attached to the central atom. To find the steric number to understand the hybridization. These electrons form the pi bonds with sulfur and are shown as the lone pairs on the sulfur atoms.Ģ. Remaining eight valence electrons are taken up by the two unused orbitals of p. These two hybrid orbitals form sigma bonds with Carbon. This hybridization is known as sp hybridization. These orbitals then combine, creating the hybrid of sp orbitals. Some solvents, waxes, and cleaners contain carbon disulfide. Carbon disulfide is used in the manufacture of viscose rayon, cellophane, carbon tetrachloride, dyes, and rubber. It is also synthesized via the catalyzed reaction between sulfur and methane at 600☌. These valence electrons that form the double bond with the Carbon atom are in 2s and 2p orbital of the Carbon atom. Carbon disulfide is a natural product of anaerobic biodegradation. Thus it takes up eight valence electrons out of 16 valence electrons. In CS2 molecule, two double bonds are formed consisting of eight valence electrons. 
It is essential to know the type of bonding in the molecule to understand its hybridization. To understand the bond formation and its type 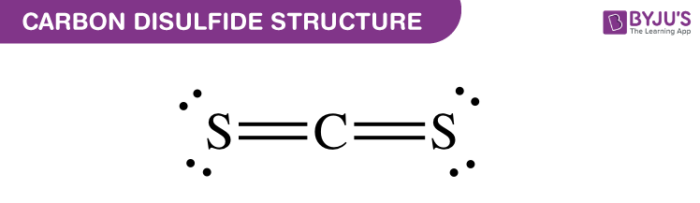
Hybridizationįor understanding the hybridization of CS2 molecule, there are two easy methods.ġ. 2 pairs of lone pairs are on each sulfur atom. Rest eight electrons are shown as lone pairs on Sulphur atom. These double bonds have eight valence electrons. These two sulfur molecules form double bonds with this Carbon molecule to complete Carbon’s octet. 
Carbon is the least electronegative moleculeand thus comes in the center. In Lewis structure of CS2 molecule, there are 16 valence electrons, out of which four valence electrons are of Carbon, and six valence electrons are from each sulfur molecule. Vapours are readily ignited and the heat of a common. Carbon disulfide has a strong disagreeable odor. It is also useful as a building block in organic chemistry. It is essential to know the Lewis structure as it helps in determining the number of lone pairs and the bond formation in the molecule. Carbon disulfide is linear in the shaping compound and used as an industrial and chemical non-polar solvent. The octet rule states that an atom should have eight electrons in its outer shell to be stable. Lewis structure follows the octet rule concept. Dots and Lines are used in this structure in which lines represent the electrons forming bonds with the central atom and dots are the non-bonding pairs. It is also used as an insecticide to kill insects on grains and fruits, or to destroy disease-causing insects and nematodes in the soil.īecause carbon disulfide dissolves organic compounds well and is undetectable by proton nuclear magnetic resonance (NMR) due to the absence of hydrogen, it is a useful solvent for NMR measurements of samples that are difficult to dissolve in heavy chloroform.Lewis structure is the structural representation of the number of valence electrons that participate in the bond formation and nonbonding electron pairs. It is also used as a vulcanization accelerator for rubber, an organic chemical raw material, and a flotation beneficiation agent. Uses of Carbon DisulfideĬarbon disulfide is mainly used as a solvent in the manufacturing process of cellophane and rayon. It is also extremely flammable and burns with a blue flame. It is insoluble in water, but is well soluble in ethanol, benzene, ether, chloroform, and carbon tetrachloride. The pure form has an ethereal, pleasant aroma, but in commercial use, often contains a foul odor due to impurities. It has a boiling point of 45.6☌ and is known for its high refractive index. It is also obtained by reacting CH4 with sulfur vapor in the presence of a catalyst. In nature, it is found in trace amounts in coal and crude oil.Ĭarbon disulfide is industrially obtained by heating charcoal and sulfur vapors. Carbon disulfide is a compound with the chemical formula CS2 and a molecular weight of 76.14. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
